 These ions and the atom of argon are known as isoelectronic.\) provides an alternative method for determining the electron configuration. Notice that the three ions have electronic configurations identical to that of inert argon. They occupy the middle portions of the long periods of the periodic table of the elements. To find valence electrons using a period table, first see if your atom is a transitional metal, which are the elements in the middle rectangle of the table. The charges on the chlorine, potassium, and calcium ions result from a strong tendency of valence electrons to adopt the stable configuration of the inert gases, with completely filled electronic shells. Transition metal, any of various chemical elements that have valence electronsi.e., electrons that can participate in the formation of chemical bondsin two shells instead of only one. Table 2 compares three ions and a neutral atom. In Table, the common oxidation numbers in the last column are interpreted as the result of either losing the valence electrons (leaving a positive ion) or gaining enough electrons to fill that valence subshell. For example, in the H 2O molecule, each H has an oxidation number of +1, and the O is –2. The group number (column number) represents the number of valence electrons common to all the elements within that group. 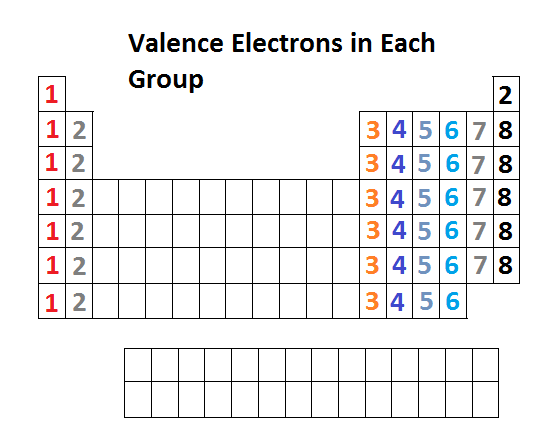
For example, the elements in the first column all have a single valence electron, an electron that can be donated in a chemical reaction. These electrons are known as valence electrons. In molecules, the various atoms are assigned chargelike values so the sum of the oxidation numbers equals the charge on the molecule. The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on. In the periodic table of the elements, elements in a single column have the same number of electrons that can participate in a chemical reaction. These are the valence electrons.įor ions, the valence equals the electrical charge. These electrons are most distant from the positive nucleus and, therefore, are most easily transferred between atoms in chemical reactions. To determine the number of valence electrons of an atom, we have to look at the periodic table and search for the elements. Groups 312 are transition metals and have 1 or 2 valence electrons. Valence electrons, which comprise the valence shell of the atom.įor brevity, many chemists record the electron configuration of an atom by giving only its outermost subshell, like 4 s 1 for potassium or 4 s 2 for calcium. The electrons in the highest numbered subshells are the Study the third column of complete electronic configurations carefully so you understand how electrons are added to the subshell of lowest energy until it reaches its capacity then the subshell of the next energy level begins to be filled. The electronic configuration of an atom is given by listing its subshells with the number of electrons in each subshell, as shown in Tableġ.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
